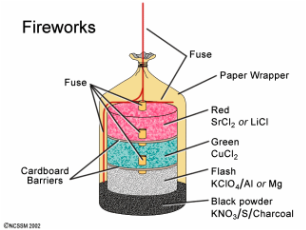
 Diagram of the Inside of a Firework.
Reference:
North Carolina School of Science and Mathematics. Fireworks. [Image on the Internet] [Date Unknown] [cited 8 November 2014]. Available from: http://www.dlt.ncssm.edu/tiger/diagrams/structure/Fireworks_CrossSection640.gif
Diagram of the Inside of a Firework.
Reference:
North Carolina School of Science and Mathematics. Fireworks. [Image on the Internet] [Date Unknown] [cited 8 November 2014]. Available from: http://www.dlt.ncssm.edu/tiger/diagrams/structure/Fireworks_CrossSection640.gif
Pros and Cons
Fireworks are used to create displays of colours and effects to entertain the public. The reaction causes lights, colours, smoke and noise to be created. This reaction can enhance celebrations, create millions of dollars for the economy, increase sales of local areas that the firework displays are occurring and it can create money from media exposure. But, fireworks also have many drawbacks, they can create fires that will cause injuries. Also, they are non-biodegradable which means they are put in landfills after use. Like all landfills, they create toxic materials that leach the ground and nearby water. Fireworks release smoke and smog that contain toxic chemicals and fumes that can damage our respiratory system. These emissions are a factor in global warming and can hurt the environment as these emissions can pollute the ecosystem and kill organisms.
Physical and Chemical Properties
Fireworks are used to create displays of colours and effects to entertain the public. The reaction causes lights, colours, smoke and noise to be created. This reaction can enhance celebrations, create millions of dollars for the economy, increase sales of local areas that the firework displays are occurring and it can create money from media exposure. But, fireworks also have many drawbacks, they can create fires that will cause injuries. Also, they are non-biodegradable which means they are put in landfills after use. Like all landfills, they create toxic materials that leach the ground and nearby water. Fireworks release smoke and smog that contain toxic chemicals and fumes that can damage our respiratory system. These emissions are a factor in global warming and can hurt the environment as these emissions can pollute the ecosystem and kill organisms.
Physical and Chemical Properties
- Carbon, a reactant, has a dull lustre, meaning it is not shiny.
- Smoke, a product, has a black or grey colour.
- Sulfur, a reactant, is flammable, meaning it has the ability to burn.
- Phosphorous, a reactant, oxidizable, meaning it is capable of reacting with oxygen.
- 10KNO3 (s) + 3S (s) + 8C (s) → 2KCO3 (s) + 3K2SO4 (s) + 6CO2 (g) + 5N2
- Combustion reaction
